Pentagonal pyramidal molecular geometry
| Pentagonal pyramidal molecular geometry | |
|---|---|
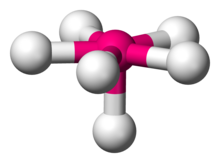 | |
| Examples | XeOF− 5 |
| Point group | C5v |
| Coordination number | 6 |
| Bond angle(s) | 90°, 72° |
| μ (Polarity) | >0 |
In chemistry, pentagonal pyramidal molecular geometry describes the shape of compounds where in six atoms or groups of atoms or ligands are arranged around a central atom, at the vertices of a pentagonal pyramid. It is one of the few molecular geometries with uneven bond angles.

Examples
References
- ^ a b Baran, E. (2000). "Mean amplitudes of vibration of the pentagonal pyramidal XeOF−
5 and IOF2−
5 anions". J. Fluorine Chem. 101: 61–63. doi:10.1016/S0022-1139(99)00194-3. - ^ Housecroft, Catherine E.; Sharpe, Alan G. (2005). Inorganic Chemistry (2nd ed.). Pearson Prentice-Hall. p. 485. ISBN 0130-39913-2.
Pentagonal pyramid, Wolfram MathWorld
- v
- t
- e
Molecular geometry
- VSEPR theory
- Coordination number
- Linear
- Bent
- Trigonal planar
- Trigonal pyramidal
- T-shaped
- Tetrahedral
- Square planar
- Seesaw
- Octahedral
- Trigonal prismatic
- Pentagonal pyramidal
 Category
Category
 | This stereochemistry article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











