ELB-139
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
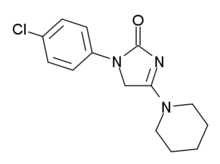
| Formula | C14H16ClN3O |
| Molar mass | 277.75 g·mol−1 |
| 3D model (JSmol) |
|
| |
InChI
| |
| (verify) | |
ELB-139 (LS-191,811) is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.[1][2]
ELB-139 is a subtype-selective partial agonist at GABAA receptors, with highest affinity for the α3 subtype, but highest efficacy at α1 and α2.[3] It has primarily anxiolytic and anticonvulsant effects, but produces little sedative effects or ataxia,[4] and has also been demonstrated in rats to increase serotonin levels in the striatum and prefrontal cortex, without affecting dopamine levels.[5] It has been proposed as a possible candidate for a novel non-sedating anxiolytic or anticonvulsant drug for use in humans[6] The sponsor elbion AG registered a clinical trial in ClinicalTrials.gov for the treatment of anxiety associated with panic disorder but the results have not been reported.[7] It was developed by Arzneimittelwerk Dresden in the 1990s.[8]
References
- ^ Langen B, Egerland U, Bernöster K, Dost R, Unverferth K, Rundfeldt C (August 2005). "Characterization in rats of the anxiolytic potential of ELB139 [1-(4-chlorophenyl)-4-piperidin-1-yl-1,5-dihydro-imidazol-2-on], a new agonist at the benzodiazepine binding site of the GABAA receptor". The Journal of Pharmacology and Experimental Therapeutics. 314 (2): 717–24. doi:10.1124/jpet.105.084681. PMID 15860576. S2CID 21967108.
- ^ Atack JR (May 2005). "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs. 14 (5): 601–18. doi:10.1517/13543784.14.5.601. PMID 15926867. S2CID 22793644.
- ^ Rabe H, Kronbach C, Rundfeldt C, Lüddens H (March 2007). "The novel anxiolytic ELB139 displays selectivity to recombinant GABA(A) receptors different from diazepam". Neuropharmacology. 52 (3): 796–801. doi:10.1016/j.neuropharm.2006.09.013. PMID 17087982. S2CID 21598180.
- ^ Grunwald C, Rundfeldt C, Lankau HJ, Arnold T, Höfgen N, Dost R, et al. (March 2006). "Synthesis, pharmacology, and structure-activity relationships of novel imidazolones and pyrrolones as modulators of GABAA receptors". Journal of Medicinal Chemistry. 49 (6): 1855–66. doi:10.1021/jm0509400. PMID 16539371.
- ^ Langen B, Rundfeldt C (January 2007). "ELB139 an agonist at the benzodiazepine binding site increases 5-HT in the striatum and prefrontal cortex of rats: a microdialysis study". Pharmacology, Biochemistry, and Behavior. 86 (1): 79–85. doi:10.1016/j.pbb.2006.12.010. PMID 17257662. S2CID 22862432.
- ^ Rogawski MA (June 2006). "Diverse mechanisms of antiepileptic drugs in the development pipeline". Epilepsy Research. 69 (3): 273–94. doi:10.1016/j.eplepsyres.2006.02.004. PMC 1562526. PMID 16621450.
- ^ Whiting PJ (February 2006). "GABA-A receptors: a viable target for novel anxiolytics?". Current Opinion in Pharmacology. 6 (1): 24–9. doi:10.1016/j.coph.2005.08.005. PMID 16359919.
- ^ US 5869481, "Anticonvulsive 1-ar(alk)ylimidazolin-2-ones and process for making"
- v
- t
- e
- Benzodiazepines: Adinazolam
- Alprazolam
- Bromazepam
- Camazepam
- Chlordiazepoxide
- Clobazam
- Clonazepam
- Clorazepate
- Clotiazepam
- Cloxazolam
- Diazepam#
- Ethyl loflazepate
- Etizolam
- Fludiazepam
- Halazepam
- Ketazolam
- Lorazepam#
- Medazepam
- Nordazepam
- Oxazepam
- Pinazepam
- Prazepam; Others: Alpidem‡
- Barbiturates (e.g., phenobarbital)
- Carbamates (e.g., meprobamate)
- Carisoprodol
- Chlormezanone‡
- Ethanol (alcohol)
- Etifoxine
(α2δ VDCC blockers)
- SSRIsTooltip Selective serotonin reuptake inhibitors (e.g., escitalopram)
- SNRIsTooltip Serotonin-norepinephrine reuptake inhibitors (e.g., duloxetine)
- SARIsTooltip Serotonin antagonist and reuptake inhibitors (e.g., trazodone)
- TCAsTooltip Tricyclic antidepressants (e.g., clomipramine#)
- TeCAsTooltip Tetracyclic antidepressants (e.g., mirtazapine)
- MAOIsTooltip Monoamine oxidase inhibitors (e.g., phenelzine); Others: Agomelatine
- Bupropion
- Tianeptine
- Vilazodone
- Vortioxetine
(Antiadrenergics)
- Alpha-1 blockers (e.g., prazosin)
- Alpha-2 agonists (e.g., clonidine, dexmedetomidine, guanfacine)
- Beta blockers (e.g., propranolol)
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
 | This article about an anxiolytic is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e










